| Realtime | Geld | Brief | Zeit |
|---|---|---|---|
| 42,950 | 43,240 | 22:29 | |
| 42,855 | 43,290 | 22:00 |
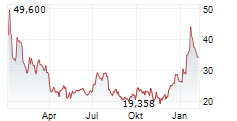
► 13.05.: | | | | | | | | |
► 12.05.: | | | | | | | | |
► Aktueller MODERNA Börsenkurs + Xetra-Orderbuch
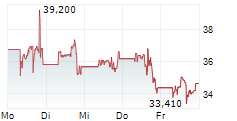
► 12.05.: | | | | | | | | |
► Aktueller MODERNA Börsenkurs + Xetra-Orderbuch
| Stück | Geld | Kurs | Brief | Stück |
|---|---|---|---|---|
| 60,00 | 15 | |||
| 57,49 | 10 | |||
| 52,60 | 100 | |||
| 52,20 | 500 | |||
| 52,00 | 44 | |||
| 51,76 | 50 | |||
| 43,460 | 172 | |||
| 43,430 | 19 | |||
| 43,315 | 20 | |||
| 43,290 | 172 | |||
Quelle: [URL] https://aktienkurs-orderbuch.finanznachrichten.de/0qf.htm [/URL] | ||||
| 172 | 42,855 | |||
| 20 | 42,760 | |||
| 17 | 42,630 | |||
| 172 | 42,440 | |||
| 100 | 41,600 | |||
| 50 | 35,000 | |||
| 55 | 26,000 | |||
| 120 | 20,800 | |||
| 1.052 | 0,000 | |||
| Summe Aktien im Kauf | Verhältnis | Summe Aktien im Verkauf | ||
| 1.758 | 0,627 | 1.102 | ||
| Uhrzeit | Aktienkurs | Stück |
|---|---|---|
| 17:35:50 | 43,310 | 1.127 |
| 17:29:32 | 43,655 | 16 |
| 17:29:32 | 43,650 | 34 |
| 17:29:16 | 43,760 | 173 |
| 17:25:54 | 43,805 | 41 |
| 17:25:46 | 43,755 | 98 |
| 17:23:32 | 43,955 | 32 |
| 17:23:32 | 43,950 | 160 |
| 17:21:24 | 43,800 | 100 |
| 17:21:24 | 43,800 | 75 |
| 17:16:03 | 43,600 | 10 |
| 17:15:03 | 43,385 | 14 |
| 17:15:03 | 43,385 | 75 |
| 17:07:05 | 43,115 | 178 |
| 17:05:23 | 43,080 | 40 |
| 16:58:40 | 43,115 | 162 |
| 16:58:40 | 43,080 | 71 |
| 16:58:40 | 43,075 | 61 |
| 16:58:31 | 43,000 | 25 |
| 16:58:27 | 43,000 | 75 |
| Tagesumsatz Xetra | -2,440 -5,33 % | 18.101 |
Angaben in grüner Schrift zeigen, dass der Aktienkurs im Vergleich zum vorherigen Kurs gestiegen ist. Angaben in roter Schrift zeigen gefallene Börsenkurse.
- Alle
- Pressemitteilungen
- Empfehlungen
- Chartanalysen
- Berichte
| Zeit | Aktuelle Nachrichten Sprache:
Alle DE EN | Leser | Medien | ||
|---|---|---|---|---|---|
| MODERNA Aktie jetzt für 0€ handeln | |||||
| Di | Moderna, Inc.: Moderna Recognized by TIME as One of the World's Most Impactful Companies | 339 | ACCESS Newswire | CAMBRIDGE, MA / ACCESS Newswire / May 12, 2026 / Moderna, Inc. (NASDAQ:MRNA) today announced it was ranked no. 1 on TIME's 2026 list of the World's Most Impactful Companies. The recognition highlights... ► Artikel lesen | |
| Di | JEFFERIES stuft Moderna auf 'Hold' | 561 | dpa-AFX-Analyser | NEW YORK (dpa-AFX Analyser) - Das Analysehaus Jefferies hat die Einstufung für Moderna auf "Hold" mit einem Kursziel von 45 US-Dollar belassen. Andrew Tsai bezog am Dienstag Stellung zur jüngsten Kursstärke... ► Artikel lesen | |
| Mo | Moderna Stock Is Surging on Hantavirus Threats. Its Chart Offers a Harsh Dose of Reality. | 37 | Barchart.com | ||
| Mo | Moderna stock soars as hantavirus fears spark outbreak trade | 22 | iNVEZZ.com | ||
| Mo | Why Shares of Moderna, Dell, and Micron Jumped | 37 | Baystreet.ca |
1 2 3 4 5 Weiter >>
456 Nachrichten in den letzten 12 Monaten
| Zeit | Aktuelle Nachrichten | Medien | ||
|---|---|---|---|---|
| 19:34 | NanoRepro: Mit Elan gestartet | boersengefluester.de | "Wir sind auf einem guten Weg", sagt Lisa Jüngst, CEO von NanoRepro, am Ende ihrer Präsentation auf der Frühjahrskonferenz in Frankfurt. Die Aufgabenstellung könnte dabei ambitionierter kaum sein. Immerhin... ► Artikel lesen | |
| 19:54 | CLERHP: Larimar Signs Agreement With Point for the Outdoor Furnishing of Residences and Shared Spaces Within the Complex | Business Wire | The proposal combines design, materials, and Mediterranean aesthetics adapted to the Caribbean environment. The project further strengthens its international positioning through collaboration... ► Artikel lesen | |
| 18:06 | Aelis Farma: Combined General Meeting of June 3, 2026: Availability of Preparatory Documents and Participation and Voting Procedures | Business Wire | Regulatory News:
Aelis Farma (ISIN: FR0014007ZB4 Ticker: AELIS), a clinical-stage biopharmaceutical company specializing in the development of treatments for brain and peripheral diseases involving... ► Artikel lesen | |
| 17:00 | NurExone: Der Übergang in die klinische Phase rückt näher | Small- & Micro Cap Investment | ||
| 15:09 | EQS-HV: aap Implantate AG: Bekanntmachung der Einberufung zur Hauptversammlung am 23.06.2026 in Berlin mit dem Ziel der europaweiten Verbreitung gemäß §121 AktG | EQS Group (DE) | EQS-News: aap Implantate AG
/ Bekanntmachung der Einberufung zur Hauptversammlung
aap Implantate AG: Bekanntmachung der Einberufung zur Hauptversammlung am 23.06.2026 in Berlin... ► Artikel lesen |
Sie erhalten auf FinanzNachrichten.de kostenlose Realtime-Aktienkurse von und sowie Kurse und Daten von ARIVA.DE AG.
Weitere Kennzahlen, Fundamentaldaten und Unternehmensinformationen zu MODERNA INC finden Sie auf Wallstreet Online.