| Realtime | Geld | Brief | Zeit |
|---|---|---|---|
| 78,95 | 79,35 | 19:51 | |
| 78,95 | 79,35 | 19:51 |
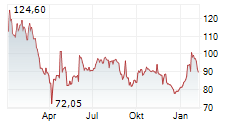
► 13.05.: | | | | | | | | |
► 12.05.: | | | | | | | | |
► Aktueller BIONTECH Börsenkurs + Xetra-Orderbuch
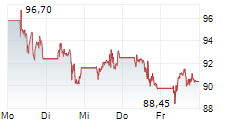
► 12.05.: | | | | | | | | |
► Aktueller BIONTECH Börsenkurs + Xetra-Orderbuch
| Stück | Geld | Kurs | Brief | Stück |
|---|---|---|---|---|
| 80,40 | 121 | |||
| 80,30 | 100 | |||
| 79,95 | 100 | |||
| 79,65 | 54 | |||
| 79,55 | 54 | |||
| 79,45 | 319 | |||
| 79,30 | 105 | |||
| 79,25 | 96 | |||
| 79,20 | 100 | |||
| 79,15 | 1.281 | |||
Quelle: [URL] https://aktienkurs-orderbuch.finanznachrichten.de/22ua.htm [/URL] | ||||
| 100 | 78,95 | |||
| 1.281 | 78,75 | |||
| 96 | 78,65 | |||
| 119 | 78,60 | |||
| 204 | 78,55 | |||
| 38 | 78,50 | |||
| 166 | 78,45 | |||
| 54 | 78,35 | |||
| 100 | 78,25 | |||
| 120 | 78,00 | |||
| Summe Aktien im Kauf | Verhältnis | Summe Aktien im Verkauf | ||
| 2.278 | 1,023 | 2.330 | ||
| Uhrzeit | Aktienkurs | Stück |
|---|---|---|
| 17:29:59 | 78,80 | 35 |
| 17:25:24 | 78,95 | 161 |
| 17:25:24 | 79,00 | 44 |
| 17:23:47 | 79,05 | 115 |
| 17:23:47 | 79,15 | 34 |
| 17:23:19 | 79,00 | 25 |
| 17:17:11 | 78,45 | 26 |
| 17:17:11 | 78,40 | 17 |
| 17:16:52 | 78,35 | 103 |
| 17:14:45 | 78,30 | 1 |
| 17:13:11 | 78,20 | 100 |
| 17:11:35 | 78,10 | 129 |
| 17:11:35 | 78,15 | 56 |
| 17:06:41 | 78,10 | 491 |
| 17:06:41 | 78,10 | 87 |
| 17:06:34 | 78,10 | 1 |
| 17:06:34 | 78,10 | 188 |
| 17:06:34 | 78,10 | 233 |
| 17:06:08 | 78,15 | 20 |
| 17:05:58 | 78,25 | 51 |
| Tagesumsatz Xetra | -0,35 -0,44 % | 28.917 |
Angaben in grüner Schrift zeigen, dass der Aktienkurs im Vergleich zum vorherigen Kurs gestiegen ist. Angaben in roter Schrift zeigen gefallene Börsenkurse.
- Alle
- Pressemitteilungen
- Empfehlungen
- Chartanalysen
- Berichte
| Zeit | Aktuelle Nachrichten Sprache:
Alle DE EN | Leser | Medien | ||
|---|---|---|---|---|---|
| 13:30 | EXKLUSIV zu Biontech: Warum erfahrene Anleger heute zugreifen - Das sollten Sie jetzt wissen | Hebelschein-Spekulant | |||
| BIONTECH Aktie jetzt für 0€ handeln | |||||
| 12:26 | BioNTech zieht Impfstoffproduktion aus Deutschland ab - bis zu 1860 Stellen betroffen | 40 | Blackout News | ||
| 09:55 | Biontech & Jumia Technologies: Handelsaussetzung - 13.05.2026 | 81 | Wiener Börse | ||
| Di | Biotech and the Efficiency Revolution: Why the Final Mile Matters Most for Novartis, BioNTech, and BioNxt | 63 | The Market Herald Canada | ||
| Di | ANALYSE-FLASH: Berenberg senkt Ziel für Biontech auf 140 Dollar - 'Buy' | 351 | dpa-AFX | HAMBURG (dpa-AFX Broker) - Die Privatbank Berenberg hat das Kursziel für Biontech von 155 auf 140 US-Dollar gesenkt, aber die Einstufung auf "Buy" belassen. Mit dem neuen Kursziel trage er dem zunehmenden... ► Artikel lesen |
1 2 3 4 5 Weiter >>
758 Nachrichten in den letzten 12 Monaten
| Zeit | Aktuelle Nachrichten | Medien | ||
|---|---|---|---|---|
| 17:00 | NurExone: Der Übergang in die klinische Phase rückt näher | Small- & Micro Cap Investment | ||
| 18:06 | Aelis Farma: Combined General Meeting of June 3, 2026: Availability of Preparatory Documents and Participation and Voting Procedures | Business Wire | Regulatory News:
Aelis Farma (ISIN: FR0014007ZB4 Ticker: AELIS), a clinical-stage biopharmaceutical company specializing in the development of treatments for brain and peripheral diseases involving... ► Artikel lesen | |
| 15:09 | EQS-HV: aap Implantate AG: Bekanntmachung der Einberufung zur Hauptversammlung am 23.06.2026 in Berlin mit dem Ziel der europaweiten Verbreitung gemäß §121 AktG | EQS Group (DE) | EQS-News: aap Implantate AG
/ Bekanntmachung der Einberufung zur Hauptversammlung
aap Implantate AG: Bekanntmachung der Einberufung zur Hauptversammlung am 23.06.2026 in Berlin... ► Artikel lesen | |
| 14:36 | Vor Biopharma: Vor Bio Reports First Quarter 2026 Financial Results and Provides Corporate Update | GlobeNewswire (Europe) | Enrollment on track for Phase 3 UPSTREAM MG trial of telitacicept in generalized myasthenia gravis patients with topline results anticipated in 1H27 Enrollment ongoing for Phase 3 UPSTREAM SjD of... ► Artikel lesen | |
| 15:09 | EQS-News: GeoVax, Inc.: GeoVax Highlights Emerging Role of Gedeptin(R) in Combination Immunotherapy Strategies Targeting Immunologically Cold Tumors | EQS Group (EN) | EQS-News: GeoVax, Inc.
/ Key word(s): Science
GeoVax Highlights Emerging Role of Gedeptin(R) in Combination Immunotherapy Strategies Targeting Immunologically Cold Tumors 13.05.2026... ► Artikel lesen |
Sie erhalten auf FinanzNachrichten.de kostenlose Realtime-Aktienkurse von und sowie Kurse und Daten von ARIVA.DE AG.
Weitere Kennzahlen, Fundamentaldaten und Unternehmensinformationen zu BIONTECH SE ADR finden Sie auf Wallstreet Online.