| Realtime | Geld | Brief | Zeit |
|---|---|---|---|
| 71,58 | 71,76 | 20:56 | |
| 71,56 | 71,78 | 20:56 |
► 13.05.: | | | | | | | | |
► 12.05.: | | | | | | | | |
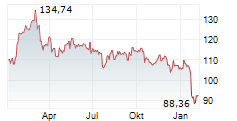
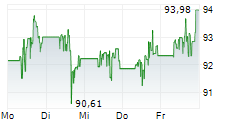
► Aktueller ABBOTT LABORATORIES Börsenkurs + Xetra-Orderbuch
► 12.05.: | | | | | | | | |
► Aktueller ABBOTT LABORATORIES Börsenkurs + Xetra-Orderbuch
| Stück | Geld | Kurs | Brief | Stück |
|---|---|---|---|---|
| 83,10 | 45 | |||
| 72,48 | 111 | |||
| 72,12 | 2 | |||
| 71,96 | 8 | |||
| 71,92 | 6 | |||
| 71,88 | 18 | |||
| 71,80 | 2 | |||
| 71,70 | 111 | |||
| 71,68 | 117 | |||
| 71,66 | 989 | |||
Quelle: [URL] https://aktienkurs-orderbuch.finanznachrichten.de/abl.htm [/URL] | ||||
| 989 | 71,52 | |||
| 111 | 71,50 | |||
| 2 | 71,32 | |||
| 2 | 71,16 | |||
| 111 | 70,92 | |||
| 50 | 70,52 | |||
| 36 | 70,00 | |||
| 40 | 68,80 | |||
| 60 | 66,20 | |||
| 50 | 65,20 | |||
| Summe Aktien im Kauf | Verhältnis | Summe Aktien im Verkauf | ||
| 1.451 | 0,971 | 1.409 | ||
| Uhrzeit | Aktienkurs | Stück |
|---|---|---|
| 17:25:46 | 71,12 | 262 |
| 16:55:07 | 71,14 | 13 |
| 16:07:24 | 71,48 | 117 |
| 15:39:28 | 70,92 | 20 |
| 15:25:57 | 71,88 | 63 |
| 14:40:34 | 71,48 | 10 |
| 14:40:34 | 71,48 | 143 |
| 14:30:11 | 71,56 | 110 |
| 13:54:03 | 71,50 | 50 |
| 12:58:24 | 71,66 | 9 |
| 12:58:24 | 71,66 | 187 |
| 12:39:24 | 72,02 | 20 |
| 12:30:00 | 71,66 | 5 |
| 12:07:38 | 71,66 | 1 |
| 11:37:15 | 71,56 | 147 |
| 11:37:15 | 71,58 | 99 |
| 11:37:04 | 71,70 | 1 |
| 11:37:04 | 71,70 | 50 |
| 11:37:04 | 71,70 | 21 |
| 11:37:04 | 71,68 | 54 |
| Tagesumsatz Xetra | -0,36 -0,50 % | 2.552 |
Angaben in grüner Schrift zeigen, dass der Aktienkurs im Vergleich zum vorherigen Kurs gestiegen ist. Angaben in roter Schrift zeigen gefallene Börsenkurse.
- Alle
- Pressemitteilungen
- Empfehlungen
- Chartanalysen
- Berichte
| Zeit | Aktuelle Nachrichten Sprache:
Alle DE EN | Leser | Medien | ||
|---|---|---|---|---|---|
| Fr | Abbott Laboratories Stock: Analyst Estimates & Ratings | 91 | Barchart.com | ||
| 05.05. | Abbott Stock Is Trading at 52-Week Lows but Insiders Are Gobbling Up Shares of This Blue-Chip Dividend Payer | 67 | Barchart.com | ||
| 05.05. | Missouri appeals court upholds $495M ruling against Abbott in baby formula case | 39 | Seeking Alpha | ||
| 05.05. | GLP-1 Hydration Products Market Outlook 2036 with Key Contributions from Smoothie King, Liquid I.V., Electrolit, Abbott, Nestlé Health Science, and ADM | Future Market Insights | 2.270 | PR Newswire | NEWARK, Del., May 5, 2026 /PRNewswire/ -- According to the latest market analysis by Future Market Insights (FMI), the global GLP-1 hydration products market is entering a rapid expansion... ► Artikel lesen | |
| ABBOTT LABORATORIES Aktie jetzt für 0€ handeln | |||||
| 02.05. | Insider trades: Abbott, UnitedHealth and ServiceNow among notable names | 128 | Seeking Alpha |
1 2 3 4 5 Weiter >>
330 Nachrichten in den letzten 12 Monaten
| Zeit | Aktuelle Nachrichten | Medien | ||
|---|---|---|---|---|
| 15:23 | DAX-Wert mit 8%-Plus: Merck KGaA hebt Jahresziele an - Aktie springt auf Zweimonatshoch | wallstreetONLINE | © Foto: Arne Dedert/dpaMerck hebt seine Jahresziele an, das Quartalsergebnis übertrifft die Erwartungen - und die Aktie legt auf einen Schlag um bis zu 8 Prozent zu. Was hinter dem überraschend starken... ► Artikel lesen | |
| 18:38 | DAX leicht erholt: E.on und Merck legen zu, Infineon, Verbio und Jenoptik zweistellig im Plus | Der Aktionär | Der DAX hat am Mittwoch einen Teil seiner kräftigen Verluste vom Vortag wettgemacht. Neben vielen Quartalszahlen richtete sich der Blick der Anleger auf den China-Besuch von US-Präsident Donald Trump.... ► Artikel lesen | |
| 18:21 | ROUNDUP/Aktien Frankfurt Schluss: Erholung - Hoffnung auf Vermittler China | dpa-AFX | FRANKFURT (dpa-AFX) - Der Dax hat sich am Mittwoch etwas von seinen deutlichen Vortagsverlusten erholt. Im Fokus stand neben einer erneuten Flut von Unternehmenszahlen die Ankunft von US-Präsident... ► Artikel lesen | |
| 15:20 | dpa-AFX-Überblick: UNTERNEHMEN vom 13.05.2026 - 15.15 Uhr | dpa-AFX | ROUNDUP 2: Merck hebt nach unerwartet starkem Start Jahresziele an - Dax-Spitze DARMSTADT - Der Merck-Konzern hat zum Jahresstart starken negativen Wechselkurseffekten und Umsatzrückgängen bei wichtigen... ► Artikel lesen | |
| 15:14 | Red Light Holland Corp.: Red Light Holland Announces Shareholder Approval of Name Change and Announces Voting Results from 2026 Annual General and Special Meeting of Shareholders | Newsfile | Toronto, Ontario--(Newsfile Corp. - May 13, 2026) - Red Light Holland Corp. (CSE: TRIP) (OTCQB: TRUFF) (FSE: 4YX) ("Red Light Holland" or the "Company") is pleased to announce that shareholders... ► Artikel lesen |
Sie erhalten auf FinanzNachrichten.de kostenlose Realtime-Aktienkurse von und sowie Kurse und Daten von ARIVA.DE AG.
Weitere Kennzahlen, Fundamentaldaten und Unternehmensinformationen zu ABBOTT LABORATORIES finden Sie auf Wallstreet Online.