| Realtime | Geld | Brief | Zeit |
|---|---|---|---|
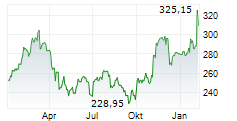
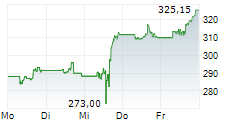
| 287,00 | 287,50 | 20:45 | |
| 286,80 | 287,75 | 20:44 |
► 13.05.: | | | | | | | | |
► 12.05.: | | | | | | | | |
► Aktueller AMGEN Börsenkurs + Xetra-Orderbuch
► 12.05.: | | | | | | | | |
► Aktueller AMGEN Börsenkurs + Xetra-Orderbuch
| Stück | Geld | Kurs | Brief | Stück |
|---|---|---|---|---|
| 349,00 | 10 | |||
| 338,10 | 1 | |||
| 287,55 | 29 | |||
| 287,30 | 272 | |||
Quelle: [URL] https://aktienkurs-orderbuch.finanznachrichten.de/amg.htm [/URL] | ||||
| 272 | 286,70 | |||
| 29 | 286,50 | |||
| 4 | 270,00 | |||
| 1.053 | 0,000 | |||
| Summe Aktien im Kauf | Verhältnis | Summe Aktien im Verkauf | ||
| 1.358 | 0,230 | 312 | ||
| Uhrzeit | Aktienkurs | Stück |
|---|---|---|
| 17:28:36 | 284,80 | 4 |
| 16:43:31 | 283,40 | 17 |
| 15:40:45 | 286,85 | 3 |
| 15:39:41 | 286,95 | 63 |
| 14:31:42 | 285,30 | 13 |
| 11:27:51 | 285,75 | 15 |
| 11:27:51 | 285,80 | 4 |
| 11:27:51 | 285,80 | 30 |
| 10:14:42 | 285,50 | 12 |
| 10:03:17 | 286,20 | 10 |
| 10:03:17 | 286,20 | 20 |
| 10:03:17 | 286,20 | 10 |
| 10:03:13 | 286,00 | 80 |
| 10:03:13 | 285,95 | 10 |
| 10:03:13 | 285,95 | 1 |
| 09:52:43 | 286,15 | 15 |
| 09:38:08 | 286,20 | 7 |
| 09:04:46 | 287,65 | 1 |
| 09:04:45 | 286,45 | 2 |
| Tagesumsatz Xetra | -2,10 -0,73 % | 476 |
Angaben in grüner Schrift zeigen, dass der Aktienkurs im Vergleich zum vorherigen Kurs gestiegen ist. Angaben in roter Schrift zeigen gefallene Börsenkurse.
- Alle
- Pressemitteilungen
- Empfehlungen
- Chartanalysen
- Berichte
| Zeit | Aktuelle Nachrichten Sprache:
Alle DE EN | Leser | Medien | ||
|---|---|---|---|---|---|
| AMGEN Aktie jetzt für 0€ handeln | |||||
| Di | Maria Elvira Salazar trades in Amgen, Carrier Global, and Microsoft stocks | 14 | Investing.com | ||
| Sa | Amgen Q1 2026: Sechs Wachstumstreiber sorgen für 24 % Expansion | 21 | Investing.com Deutsch | ||
| Sa | Amgen Q1 2026 slides: six growth drivers fuel 24% expansion | 10 | Investing.com | ||
| Sa | Amgen übertrifft Gewinnprognose für Q1 2026, Aktie gibt dennoch nach | 18 | Investing.com Deutsch | ||
| Do | Freedom Broker stuft Amgen auf 'Kaufen' hoch: Wachstumstreiber intakt | 20 | Investing.com Deutsch |
1 2 3 4 5 Weiter >>
338 Nachrichten in den letzten 12 Monaten
| Zeit | Aktuelle Nachrichten | Medien | ||
|---|---|---|---|---|
| 19:34 | NanoRepro: Mit Elan gestartet | boersengefluester.de | "Wir sind auf einem guten Weg", sagt Lisa Jüngst, CEO von NanoRepro, am Ende ihrer Präsentation auf der Frühjahrskonferenz in Frankfurt. Die Aufgabenstellung könnte dabei ambitionierter kaum sein. Immerhin... ► Artikel lesen | |
| 17:00 | NurExone: Der Übergang in die klinische Phase rückt näher | Small- & Micro Cap Investment | ||
| 18:06 | Aelis Farma: Combined General Meeting of June 3, 2026: Availability of Preparatory Documents and Participation and Voting Procedures | Business Wire | Regulatory News:
Aelis Farma (ISIN: FR0014007ZB4 Ticker: AELIS), a clinical-stage biopharmaceutical company specializing in the development of treatments for brain and peripheral diseases involving... ► Artikel lesen | |
| 15:09 | EQS-HV: aap Implantate AG: Bekanntmachung der Einberufung zur Hauptversammlung am 23.06.2026 in Berlin mit dem Ziel der europaweiten Verbreitung gemäß §121 AktG | EQS Group (DE) | EQS-News: aap Implantate AG
/ Bekanntmachung der Einberufung zur Hauptversammlung
aap Implantate AG: Bekanntmachung der Einberufung zur Hauptversammlung am 23.06.2026 in Berlin... ► Artikel lesen | |
| 14:36 | Vor Biopharma: Vor Bio Reports First Quarter 2026 Financial Results and Provides Corporate Update | GlobeNewswire (Europe) | Enrollment on track for Phase 3 UPSTREAM MG trial of telitacicept in generalized myasthenia gravis patients with topline results anticipated in 1H27 Enrollment ongoing for Phase 3 UPSTREAM SjD of... ► Artikel lesen |
Sie erhalten auf FinanzNachrichten.de kostenlose Realtime-Aktienkurse von und sowie Kurse und Daten von ARIVA.DE AG.
Weitere Kennzahlen, Fundamentaldaten und Unternehmensinformationen zu AMGEN INC finden Sie auf Wallstreet Online.