| Realtime | Geld | Brief | Zeit |
|---|---|---|---|
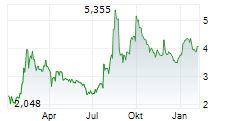
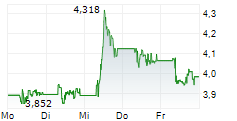
| 2,539 | 2,573 | 22:19 | |
| 2,534 | 2,572 | 22:00 |
► 13.05.: | | | | | | | | |
► 12.05.: | | | | | | | | |
► Aktueller VALNEVA Börsenkurs + Xetra-Orderbuch
► 12.05.: | | | | | | | | |
► Aktueller VALNEVA Börsenkurs + Xetra-Orderbuch
| Stück | Geld | Kurs | Brief | Stück |
|---|---|---|---|---|
| 9,990 | 1.400 | |||
| 9,900 | 100 | |||
| 8,500 | 655 | |||
| 7,250 | 5.000 | |||
| 5,500 | 2.000 | |||
| 5,100 | 200 | |||
| 4,000 | 500 | |||
| 3,600 | 3.000 | |||
| 2,647 | 3.200 | |||
| 2,572 | 3.200 | |||
Quelle: [URL] https://aktienkurs-orderbuch.finanznachrichten.de/ayj.htm [/URL] | ||||
| 3.200 | 2,534 | |||
| 3.200 | 2,485 | |||
| 10.000 | 2,250 | |||
| 100 | 2,200 | |||
| 300 | 1,700 | |||
| 1.000 | 0,037 | |||
| 1.068 | 0,000 | |||
| Summe Aktien im Kauf | Verhältnis | Summe Aktien im Verkauf | ||
| 18.868 | 1,021 | 19.255 | ||
| Uhrzeit | Aktienkurs | Stück |
|---|---|---|
| 17:29:18 | 2,597 | 57 |
| 17:29:09 | 2,603 | 53 |
| 17:29:09 | 2,603 | 87 |
| 17:29:09 | 2,603 | 107 |
| 17:19:59 | 2,616 | 88 |
| 16:55:10 | 2,625 | 2.288 |
| 16:55:10 | 2,625 | 1.263 |
| 16:26:35 | 2,616 | 11 |
| 16:01:18 | 2,622 | 71 |
| 15:01:28 | 2,595 | 550 |
| 15:01:28 | 2,595 | 218 |
| 13:22:22 | 2,535 | 1.000 |
| 13:22:22 | 2,535 | 10 |
| 13:17:40 | 2,517 | 330 |
| 13:11:23 | 2,560 | 2.021 |
| 13:11:23 | 2,560 | 2.254 |
| 13:11:23 | 2,562 | 500 |
| 12:11:41 | 2,572 | 74 |
| 12:11:41 | 2,572 | 250 |
| 12:11:41 | 2,564 | 707 |
| Tagesumsatz Xetra | +0,001 +0,04 % | 92.072 |
Angaben in grüner Schrift zeigen, dass der Aktienkurs im Vergleich zum vorherigen Kurs gestiegen ist. Angaben in roter Schrift zeigen gefallene Börsenkurse.
- Alle
- Pressemitteilungen
- Empfehlungen
- Chartanalysen
- Berichte
| Zeit | Aktuelle Nachrichten Sprache:
Alle DE EN | Leser | Medien | ||
|---|---|---|---|---|---|
| 17:54 | Transcript: Valneva Q1 2026 Earnings Conference Call | 22 | Benzinga.com | ||
| 15:26 | Valneva to lay off up to 15% of workforce in face of 'adverse trend' in travel vaccines | 39 | FierceBiotech | ||
| 13:30 | Valneva im Blickpunkt: Warum Insider bereits massiv kaufen - Was ich meinen Lesern jetzt rate | Hebelschein-Spekulant | |||
| 11:15 | Valneva senkt Prognose und baut Stellen ab | 520 | 4investors | Beim Impfstoffspezialisten Valneva SE häufen sich im ersten Quartal 2026 die Belastungen: Der Nettoverlust weitete sich auf 32,1 Millionen Euro nach 9,2 Millionen Euro im Vorjahr aus, das bereinigte... ► Artikel lesen | |
| VALNEVA Aktie jetzt für 0€ handeln | |||||
| 07:18 | Valneva GAAP EPS of -€0.18, revenue of €30.9M; updates FY26 outlook | 111 | Seeking Alpha |
1 2 3 4 5 Weiter >>
190 Nachrichten in den letzten 12 Monaten
| Zeit | Aktuelle Nachrichten | Medien | ||
|---|---|---|---|---|
| 19:34 | NanoRepro: Mit Elan gestartet | boersengefluester.de | "Wir sind auf einem guten Weg", sagt Lisa Jüngst, CEO von NanoRepro, am Ende ihrer Präsentation auf der Frühjahrskonferenz in Frankfurt. Die Aufgabenstellung könnte dabei ambitionierter kaum sein. Immerhin... ► Artikel lesen | |
| 19:54 | CLERHP: Larimar Signs Agreement With Point for the Outdoor Furnishing of Residences and Shared Spaces Within the Complex | Business Wire | The proposal combines design, materials, and Mediterranean aesthetics adapted to the Caribbean environment. The project further strengthens its international positioning through collaboration... ► Artikel lesen | |
| 18:06 | Aelis Farma: Combined General Meeting of June 3, 2026: Availability of Preparatory Documents and Participation and Voting Procedures | Business Wire | Regulatory News:
Aelis Farma (ISIN: FR0014007ZB4 Ticker: AELIS), a clinical-stage biopharmaceutical company specializing in the development of treatments for brain and peripheral diseases involving... ► Artikel lesen | |
| 17:00 | NurExone: Der Übergang in die klinische Phase rückt näher | Small- & Micro Cap Investment | ||
| 15:09 | EQS-HV: aap Implantate AG: Bekanntmachung der Einberufung zur Hauptversammlung am 23.06.2026 in Berlin mit dem Ziel der europaweiten Verbreitung gemäß §121 AktG | EQS Group (DE) | EQS-News: aap Implantate AG
/ Bekanntmachung der Einberufung zur Hauptversammlung
aap Implantate AG: Bekanntmachung der Einberufung zur Hauptversammlung am 23.06.2026 in Berlin... ► Artikel lesen |
Sie erhalten auf FinanzNachrichten.de kostenlose Realtime-Aktienkurse von und sowie Kurse und Daten von ARIVA.DE AG.
Weitere Kennzahlen, Fundamentaldaten und Unternehmensinformationen zu VALNEVA SE finden Sie auf Wallstreet Online.