| Realtime | Geld | Brief | Zeit |
|---|---|---|---|
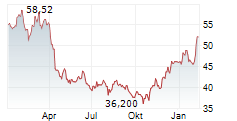
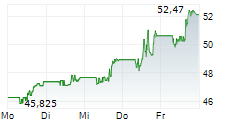
| 47,850 | 48,385 | 13.05. | |
| 48,000 | 48,340 | 13.05. |
► 13.05.: | | | | | | | | |
► 12.05.: | | | | | | | | | 17:30
► Aktueller BRISTOL-MYERS SQUIBB Börsenkurs + Xetra-Orderbuch
► 12.05.: | | | | | | | | | 17:30
► Aktueller BRISTOL-MYERS SQUIBB Börsenkurs + Xetra-Orderbuch
| Stück | Geld | Kurs | Brief | Stück |
|---|---|---|---|---|
| 49,455 | 178 | |||
| 48,505 | 169 | |||
| 48,335 | 1 | |||
| 48,195 | 9 | |||
| 48,185 | 9 | |||
| 48,175 | 18 | |||
| 48,145 | 1 | |||
| 48,065 | 168 | |||
| 48,050 | 178 | |||
| 48,030 | 1.794 | |||
Quelle: [URL] https://aktienkurs-orderbuch.finanznachrichten.de/brm.htm [/URL] | ||||
| 1.616 | 47,870 | |||
| 168 | 47,860 | |||
| 1 | 47,630 | |||
| 1 | 47,460 | |||
| 168 | 47,450 | |||
| 178 | 47,010 | |||
| 100 | 47,000 | |||
| 20 | 45,000 | |||
| 110 | 36,600 | |||
| 1.093 | 0,000 | |||
| Summe Aktien im Kauf | Verhältnis | Summe Aktien im Verkauf | ||
| 3.455 | 0,731 | 2.525 | ||
| Uhrzeit | Aktienkurs | Stück |
|---|---|---|
| 17:28:59 | 48,060 | 1 |
| 17:27:30 | 48,070 | 12 |
| 17:15:39 | 47,730 | 10 |
| 15:38:02 | 48,125 | 100 |
| 15:26:58 | 48,250 | 20 |
| 10:11:02 | 47,810 | 14 |
| 10:11:02 | 47,890 | 41 |
| 10:11:02 | 47,945 | 13 |
| 10:11:02 | 48,035 | 51 |
| 10:11:02 | 48,040 | 172 |
| Tagesumsatz Xetra | -0,230 -0,48 % | 533 |
Angaben in grüner Schrift zeigen, dass der Aktienkurs im Vergleich zum vorherigen Kurs gestiegen ist. Angaben in roter Schrift zeigen gefallene Börsenkurse.
- Alle
- Pressemitteilungen
- Empfehlungen
- Chartanalysen
- Berichte
| Zeit | Aktuelle Nachrichten Sprache:
Alle DE EN | Leser | Medien | ||
|---|---|---|---|---|---|
| BRISTOL-MYERS SQUIBB Aktie jetzt für 0€ handeln | |||||
| Mi | Bernstein reiterates Bristol-Myers Squibb stock rating on China deal | 6 | Investing.com | ||
| Mi | Bristol Myers Squibb And Hengrui Pharma Enter $15.2 Billion Global Collaboration To Advance 13 Early-Stage Programs | 3 | pulse2.com | ||
| Di | Bristol Myers, Hengrui Pharma Team Up in Global Drug Research Pact That Could Top $15B | 2 | MedCity News | ||
| Di | BMO bestätigt Rating für Bristol-Myers Squibb nach Milliardendeal mit Hengrui | 15 | Investing.com Deutsch | ||
| Di | BMO reiterates Bristol-Myers Squibb stock rating on Hengrui deal | 3 | Investing.com |
1 2 3 4 5 Weiter >>
425 Nachrichten in den letzten 12 Monaten
| Zeit | Aktuelle Nachrichten | Medien | ||
|---|---|---|---|---|
| Mi | DAX leicht erholt: E.on und Merck legen zu, Infineon, Verbio und Jenoptik zweistellig im Plus | Der Aktionär | Der DAX hat am Mittwoch einen Teil seiner kräftigen Verluste vom Vortag wettgemacht. Neben vielen Quartalszahlen richtete sich der Blick der Anleger auf den China-Besuch von US-Präsident Donald Trump.... ► Artikel lesen | |
| Mi | ROUNDUP/Aktien Frankfurt Schluss: Erholung - Hoffnung auf Vermittler China | dpa-AFX | FRANKFURT (dpa-AFX) - Der Dax hat sich am Mittwoch etwas von seinen deutlichen Vortagsverlusten erholt. Im Fokus stand neben einer erneuten Flut von Unternehmenszahlen die Ankunft von US-Präsident... ► Artikel lesen | |
| Mi | DAX-Wert mit 8%-Plus: Merck KGaA hebt Jahresziele an - Aktie springt auf Zweimonatshoch | wallstreetONLINE | © Foto: Arne Dedert/dpaMerck hebt seine Jahresziele an, das Quartalsergebnis übertrifft die Erwartungen - und die Aktie legt auf einen Schlag um bis zu 8 Prozent zu. Was hinter dem überraschend starken... ► Artikel lesen | |
| Mi | dpa-AFX-Überblick: UNTERNEHMEN vom 13.05.2026 - 15.15 Uhr | dpa-AFX | ROUNDUP 2: Merck hebt nach unerwartet starkem Start Jahresziele an - Dax-Spitze DARMSTADT - Der Merck-Konzern hat zum Jahresstart starken negativen Wechselkurseffekten und Umsatzrückgängen bei wichtigen... ► Artikel lesen | |
| Mi | JEFFERIES stuft ASTRAZENECA auf 'Buy' | dpa-AFX-Analyser | NEW YORK (dpa-AFX Analyser) - Das Analysehaus Jefferies hat die Einstufung für Astrazeneca auf "Buy" mit einem Kursziel von 18000 Pence belassen. Michael Leuchten attestierte dem Pharmakonzern in einer... ► Artikel lesen |
Sie erhalten auf FinanzNachrichten.de kostenlose Realtime-Aktienkurse von und sowie Kurse und Daten von ARIVA.DE AG.
Weitere Kennzahlen, Fundamentaldaten und Unternehmensinformationen zu BRISTOL-MYERS SQUIBB COMPANY finden Sie auf Wallstreet Online.