| Realtime | Geld | Brief | Zeit |
|---|---|---|---|
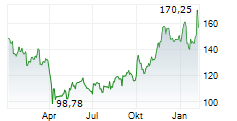
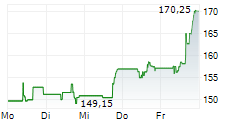
| 172,74 | 173,98 | 10:31 | |
| 172,74 | 173,98 | 10:31 |
► 14.05.: | |
11:00 |
12:00 |
12:55 |
14:00 |
15:00 |
16:00 |
17:00 |
17:30
► 13.05.: | | | | | | | | |
► Aktueller BIOGEN Börsenkurs + Xetra-Orderbuch
► 13.05.: | | | | | | | | |
► Aktueller BIOGEN Börsenkurs + Xetra-Orderbuch
| Stück | Geld | Kurs | Brief | Stück |
|---|---|---|---|---|
| 182,36 | 1.100 | |||
| 181,42 | 1.100 | |||
| 179,72 | 114 | |||
| 179,54 | 1.100 | |||
| 176,86 | 10 | |||
| 176,80 | 300 | |||
| 176,42 | 114 | |||
| 174,88 | 15 | |||
| 174,86 | 40 | |||
| 174,84 | 80 | |||
Quelle: [URL] https://aktienkurs-orderbuch.finanznachrichten.de/idp.htm [/URL] | ||||
| 45 | 166,86 | |||
| 51 | 166,84 | |||
| 51 | 166,80 | |||
| 114 | 165,34 | |||
| 300 | 164,52 | |||
| 51 | 163,76 | |||
| 1.100 | 162,80 | |||
| 114 | 162,16 | |||
| 22 | 148,00 | |||
| 40 | 1,234 | |||
| Summe Aktien im Kauf | Verhältnis | Summe Aktien im Verkauf | ||
| 1.888 | 2,104 | 3.973 | ||
| Uhrzeit | Aktienkurs | Stück |
|---|---|---|
| 10:10:05 | 171,36 | 60 |
| 09:08:06 | 174,02 | 6 |
| 09:08:06 | 174,02 | 24 |
| 09:05:53 | 174,02 | 50 |
| 09:04:39 | 173,34 | 6 |
| Tagesumsatz Xetra | +0,64 +0,37 % | 146 |
Angaben in grüner Schrift zeigen, dass der Aktienkurs im Vergleich zum vorherigen Kurs gestiegen ist. Angaben in roter Schrift zeigen gefallene Börsenkurse.
- Alle
- Pressemitteilungen
- Empfehlungen
- Chartanalysen
- Berichte
| Zeit | Aktuelle Nachrichten Sprache:
Alle DE EN | Leser | Medien | ||
|---|---|---|---|---|---|
| 09:50 | IDEC Corp Reveals Rise In Full Year Bottom Line | 2 | RTTNews | ||
| Mi | 1:6 Bonus Issue: Biogen Pharmachem stock hits 52-week low despite shareholder reward; check record date | 4 | ZEE Business | ||
| So | Apellis legt Zahlen vor - letzter Bericht vor Abschluss des Biogen-Deals | 14 | Investing.com Deutsch | ||
| BIOGEN Aktie jetzt für 0€ handeln | |||||
| Fr | Biogen Stock Gains Despite Extended FDA Review For Alzheimer's Therapy | 8 | Benzinga.com | ||
| Fr | Eisai, Biogen face delay to subcutaneous Leqembi | 2 | pharmaphorum |
1 2 3 4 5 Weiter >>
321 Nachrichten in den letzten 12 Monaten
| Zeit | Aktuelle Nachrichten | Medien | ||
|---|---|---|---|---|
| 05:10 | Schachmatt für den Krebs: Was Eli Lilly und Bayer von Vidac Pharma lernen können | inv3st.de | Die moderne Medizin steht vor wegweisenden Innovationen, bei denen die Steuerung des zellulären Energiehaushalts - die sogenannte metabolische Korrektur - zur entscheidenden Strategie wird. Während... ► Artikel lesen | |
| 06:46 | Bayer und BASF im Performance-Rausch und MustGrow Biologics als Geheimwaffe gegen den Welthunger! | zukunftsbilanzen.de | Erstellt und veröffentlicht im Auftrag von MustGrow Biologics Corp. Die Weltbevölkerung wächst unaufhaltsam, und mit ihr der Hunger nach effizienten, aber vor allem nachhaltigen Lösungen in der Landwirtschaft.... ► Artikel lesen | |
| Mi | NanoRepro: Mit Elan gestartet | boersengefluester.de | "Wir sind auf einem guten Weg", sagt Lisa Jüngst, CEO von NanoRepro, am Ende ihrer Präsentation auf der Frühjahrskonferenz in Frankfurt. Die Aufgabenstellung könnte dabei ambitionierter kaum sein. Immerhin... ► Artikel lesen | |
| 09:15 | Aktien im Fokus - Deutsche Telekom: Jetzt oder nie - DEFAMA, Evotec, SFC Energy, Valneva und Verbio | 4investors | Wo ist am Aktienmarkt etwas los, welche Themen interessieren Anleger derzeit besonders? Vor allem für Trader ist es wichtig zu wissen, wo "die Musik spielt" und welche Themen an der Börse aktuell besonders... ► Artikel lesen | |
| 09:10 | Evotec-Aktie mit -11% Einbruch - Panik nimmt weiter zu | sharedeals.de | Die Evotec-Aktie ist gestern -10,63% eingebrochen und geriet damit erneut massiv unter Druck. Nach einer zuvor kurzfristigen Stabilisierung verschlechterte sich das Chartbild wieder, während wichtige... ► Artikel lesen |
Sie erhalten auf FinanzNachrichten.de kostenlose Realtime-Aktienkurse von und sowie Kurse und Daten von ARIVA.DE AG.
Weitere Kennzahlen, Fundamentaldaten und Unternehmensinformationen zu BIOGEN INC finden Sie auf Wallstreet Online.