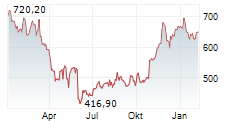
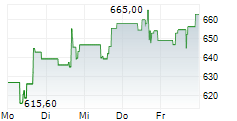
| Realtime | Geld | Brief | Zeit |
|---|---|---|---|
| 549,50 | 551,20 | 18:17 | |
| 549,20 | 551,30 | 18:16 |
► 19.05.: | | | | | | | | |
► 18.05.: | | | | | | | | |
► Aktueller REGENERON PHARMACEUTICALS Börsenkurs + Xetra-Orderbuch
► 18.05.: | | | | | | | | |
► Aktueller REGENERON PHARMACEUTICALS Börsenkurs + Xetra-Orderbuch
| Stück | Geld | Kurs | Brief | Stück |
|---|---|---|---|---|
| 548,70 | 104 | |||
Quelle: [URL] https://aktienkurs-orderbuch.finanznachrichten.de/rgo.htm [/URL] | ||||
| 104 | 547,60 | |||
| Summe Aktien im Kauf | Verhältnis | Summe Aktien im Verkauf | ||
| 104 | 1,000 | 104 | ||
| Uhrzeit | Aktienkurs | Stück |
|---|---|---|
| 13:10:28 | 526,00 | 8 |
| Tagesumsatz Xetra | -107,80 -17,01 % | 154 |
Angaben in grüner Schrift zeigen, dass der Aktienkurs im Vergleich zum vorherigen Kurs gestiegen ist. Angaben in roter Schrift zeigen gefallene Börsenkurse.
- Alle
- Pressemitteilungen
- Empfehlungen
- Chartanalysen
- Berichte
| Zeit | Aktuelle Nachrichten Sprache:
Alle DE EN | Leser | Medien | ||
|---|---|---|---|---|---|
| 13:30 | Canaccord lowers Regeneron stock price target on pipeline concerns | 5 | Investing.com | ||
| 11:46 | Regeneron signs $2.32bn deal with Parabilis for Helicon conjugates | 5 | Pharmaceutical Technology | ||
| Mo | Wolfe Research lowers Regeneron stock price target to $860 on LAG-3 miss | 11 | Investing.com | ||
| REGENERON PHARMACEUTICALS Aktie jetzt für 0€ handeln | |||||
| Mo | Regeneron Sinks 10% on Failed Phase 3 Melanoma Trial. What Comes Next for REGN Stock. | 9 | Barchart.com | ||
| Mo | Regeneron stock drops double digits on oncology trial failure | 3 | iNVEZZ.com |
1 2 3 4 5 Weiter >>
371 Nachrichten in den letzten 12 Monaten
| Zeit | Aktuelle Nachrichten | Medien | ||
|---|---|---|---|---|
| 13:10 | Tiziana Life Sciences Ltd.: Tiziana Announces New Positive Clinical Data for Intranasal Foralumab in Non-Active Secondary Progressive Multiple Sclerosis | GlobeNewswire (Europe) | Favourable trends seen in stability of disability and clinically meaningful improvements in fatigue.
BOSTON, May 19, 2026 (GLOBE NEWSWIRE) -- Tiziana Life Sciences, Ltd. (Nasdaq: TLSA) ("Tiziana")... ► Artikel lesen | |
| 14:26 | Protagenic Therapeutics, Inc.: Protagenic Therapeutics Engages Floyd Regulatory Strategic Consulting as Regulatory Affairs Advisor and Danforth Health as Strategic Operations Partner | ACCESS Newswire | NEW YORK, NY / ACCESS Newswire / May 19, 2026 / Protagenic Therapeutics, Inc. (OTCQB:PTIX), a clinical-stage biopharmaceutical company developing novel peptide-based therapies for stress-related neuropsychiatric... ► Artikel lesen | |
| 12:06 | Stryker Buys Amplitude Vascular Systems For Upto $835 Mln To Expand Vascular Treatment Business | AFX News | WASHINGTON (dpa-AFX) - Tuesday, BioStar Capital announced that Amplitude Vascular Systems, Inc., a medical technology company, has been acquired by Stryker Corporation (SYK) at a purchase price... ► Artikel lesen | |
| 13:10 | OKYO Pharma LTD: OKYO Expands Scientific Advisory Board with the Appointment of International Expert Marta Sacchetti, MD, PhD | GlobeNewswire (Europe) | Dr. Sacchetti brings international expertise in clinical and translational research on ocular inflammatory and degenerative diseaseOKYO's introduction to the broader ophthalmology community at ASCRS... ► Artikel lesen | |
| 17:27 | EQS-PVR: Evotec SE: Release according to Article 40, Section 1 of the WpHG [the German Securities Trading Act] with the objective of Europe-wide distribution | EQS Group (EN) | EQS Voting Rights Announcement: Evotec SE
Evotec SE: Release according to Article 40, Section 1 of the WpHG [the German Securities Trading Act] with the objective of Europe-wide... ► Artikel lesen |
Sie erhalten auf FinanzNachrichten.de kostenlose Realtime-Aktienkurse von und sowie Kurse und Daten von ARIVA.DE AG.
Weitere Kennzahlen, Fundamentaldaten und Unternehmensinformationen zu REGENERON PHARMACEUTICALS INC finden Sie auf Wallstreet Online.